about harvi
Daniel Burkhoff MD PhD
Daniel Burkhoff, MD, PhD, is Director of Heart Failure, Hemodynamics and Mechanical Circulatory Support Research at the Cardiovascular Research Foundation and an Adjunct Associate Professor of Medicine at Columbia University in the City of New York. He holds degrees in Applied and Engineering Physics (BS, Cornell University), a medical degree (The Johns Hopkins Medical School) and a doctorate degree in Biomedical Engineering (The Johns Hopkins University). He has published over 230 peer reviewed articles in the fields of ventricular mechanics, heart failure, medical devices and cardiovascular modeling.

Marc L Dickstein MD
Marc Dickstein is a Professor of Anesthesiology in the Division of Cardiothoracic Anesthesiology at Columbia University Medical Center in New York. After completing his undergraduate studies at the University of Pennsylvania, he earned his medical degree and completed his residency and fellowship in Cardiothoracic Anesthesiology at Columbia University. He is currently a full-time faculty member in the Division of Cardiothoracic Anesthesiology at NewYork-Presbyterian Hospital in New York City. His clinical practice includes the management of patients on cardiopulmonary bypass, ECMO, LVADs, and other forms of mechanical circulatory support (MCS). His research interests include ventricular physiology, right ventricular failure, and MCS. He is passionate about teaching and has received numerous teaching awards, including the Columbia University Presidential Award for Outstanding Teaching and the Association of Academic Anesthesiologists Education Innovation Award.

One unique aspect of this Textbook is the incorporation of a relatively sophisticated, real-time model of the cardiovascular system that is used to explain the dynamic interactions between heart and vasculature. Elements of the model pop up during Try it Now options in which specific parameter values can be varied and effects observed. Use of the model is required to complete many of the Tutorials. In addition, the entire simulation can be called by returning to the Main Menu and tapping the simulation icon. So, exactly how is the model constructed?
Hydraulic networks (like the circulatory system) can be represented by electronic circuits because of the analogies between flow of fluids through pipes and flow of electrical currents through conducting wires, between pressure and voltage, between hydraulic and electrical resistance, and between hydraulic and electrical capacitance. One electrical representation of the cardiovascular system is depicted in the following Figure.
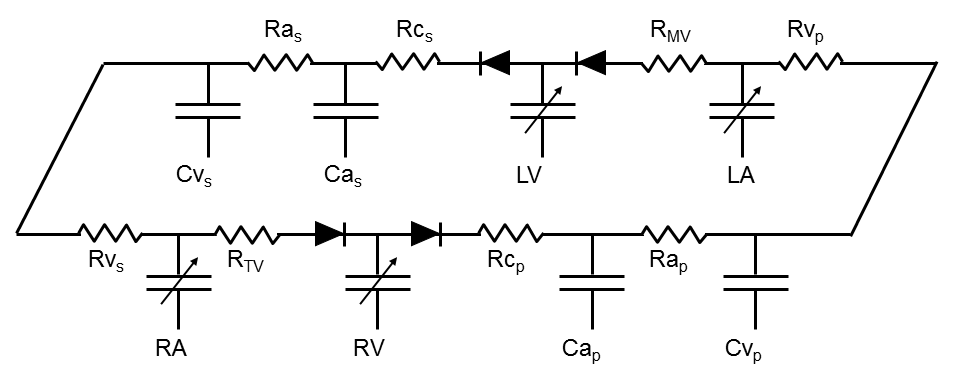
The atria and ventricles are represented by time-varying capacitances (shown by capacitors with the arrows through them). Each vascular system is represented by a series of resistances and capacitors. The heart valves are represented by diodes that permit flow in only one direction.
Using this model as a guide, a set of time varying, simultaneous differential equations can be derived to represent the circulatory system. These equations are solved in real-time and the results yield real time pressure, volume and flow tracings that form the basis of the simulations used in this textbook. More complex models of the cardiovascular system are available. However, it is remarkable how much circulatory physiology and pathophysiology this simple model explains. Of course, it is not necessary to have any understanding of electronic circuitry or differential equations to understand the remainder of this text; all explanations are based on descriptions of physiological principles without detailed reference to this model.
In addition to the basic model shown above, more complex situations can be modeled. Two examples are shunts between the atria (atrial septal defect) and between the ventricles (ventricular septal defects). The corresponding models are shown in the figures below.
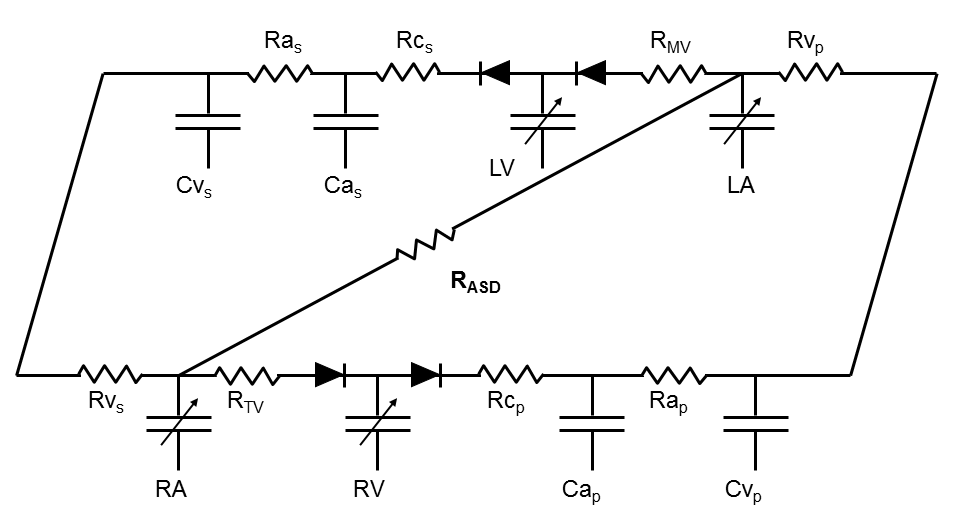

Other examples of advanced model features include the ability to introduce mechanical circulatory support devices (whose inlet and outlet sites can be selected) and the ability to simulate regurgitant and stenotic lesions into any one of the cardiac valves.
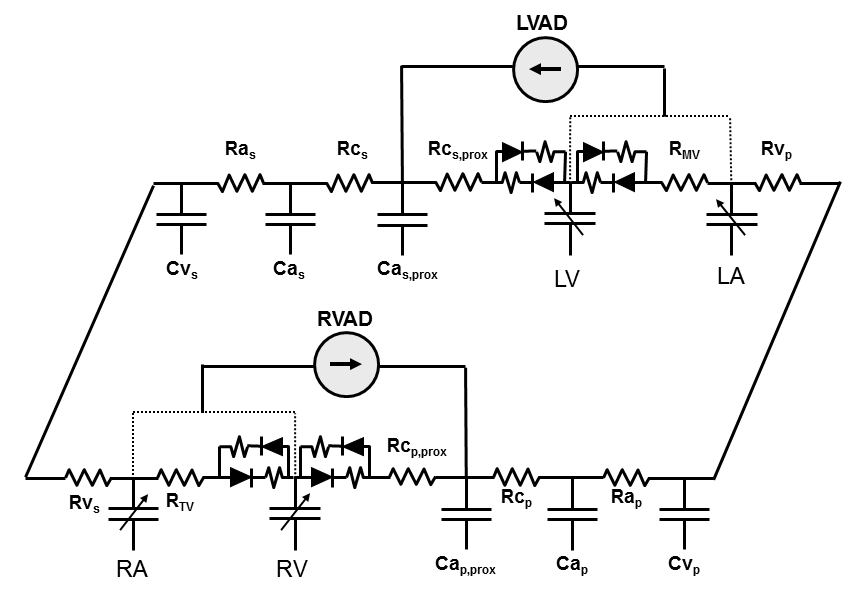
In addition to these types of model enhancements, other advanced features are incorporated into the model, such as heart cycle-dependent contractility, a pericardium, coronary vasculature, myocardial work-oxygen consumption relations and effects of simple cardiovascular drugs. These features are described in detail in various textbook chapters.
Frank O. Zur Dynamik des Herzmuskels. Z Biol 1895; 32:370-447.
Frank O. Wie Grundform des Arteriellen Pulses. Zeitschrift fGr Biologie 1899; 37:483-526.
Frank O. On the dynamics of cardiac muscle (Translated by CB Chapman and E Wasserman). Am Heart J 1959; 58:282-317.
Frank O. The basic shape of the arterial pulse. First treatise: mathematical analysis. 1899. J Mol Cell Cardiol 1990; 22:255-77.
Patterson SW, Starling EH. On the mechanical factors which determine the output of the ventricles. J Physiol 1914; 48:357-79.
Starling EH. The Linacre lecture on the law of the heart. London: Longmans, Green, 1918.
WOMERSLEY JR. Oscillatory flow in arteries: the constrained elastic tube as a model of arterial flow and pulse transmission. Phys Med Biol 1957; 2:178-87.
Guyton AC. Determination of cardiac output by equating venous return curves with cardiac response curves. Physiol Rev 1955; 35:123-9.
Guyton AC, Coleman TG, Granger HJ. Circulation: overall regulation. Annu Rev Physiol 1972; 34:13-46.
Hester RL, Brown AJ, Husband L, et al. HumMod: A Modeling Environment for the Simulation of Integrative Human Physiology. Front Physiol 2011; 2:12.
Guyton, A. C. Guyton model. Available Online at http:// www.umc.edu/Education/Schools/Medicine/Basic_Science/ Physiology_and_Biophysics/Research(Physiology)/Research. aspx. Last viewed on
Krishnamurthy A, Villongco CT, Chuang J, et al. PatientSpecific Models of Cardiac Biomechanics. J Comput Phys 2013; 244:4-21.
Smith NP, Hunter PJ, Paterson DJ. The Cardiac Physiome: at the heart of coupling models to measurement. Exp Physiol 2009; 94:469-71.
Sugiura S, Washio T, Hatano A, Okada J, Watanabe H, Hisada T. Multi-scale simulations of cardiac electrophysiology and mechanics using the University of Tokyo heart simulator. Prog Biophys Mol Biol 2012; 110:380-9.
Krenz GS, Linehan JH, Dawson CA. A fractal continuum model of the pulmonary arterial tree. J Appl Physiol (1985 ) 1992; 72:2225-37.
Krenz GS, Dawson CA. Flow and pressure distributions in vascular networks consisting of distensible vessels. Am J Physiol Heart Circ Physiol 2003; 284:H2192-H2203.
Taylor CA, Fonte TA, Min JK. Computational fluid dynamics applied to cardiac computed tomography for noninvasive quantification of fractional flow reserve: scientific basis. J Am Coll Cardiol 2013; 61:2233-41.
Zarins CK, Taylor CA, Min JK. Computed fractional flow reserve (FFTCT) derived from coronary CT angiography. J Cardiovasc Transl Res 2013; 6:708-14.
Sunagawa K, Maughan WL, Burkhoff D, Sagawa K. Left ventricular interaction with arterial load studied in isolated canine ventricle. Am J Physiol 1983; 245 (HCP 14):H773-H780.
Santamore WP, Burkhoff D. Hemodynamic consequences of ventricular interaction as assessed by model analysis. Am J Physiol 1991; 260 (HCP 29):H146-H157.
Burkhoff D, Tyberg JV. Why does pulmonary venous pressure rise following the onset of left ventricular dysfunction: a theoretical analysis. Am J Physiol 1993; 265 (HCP 34):H1819-H1828.
Guyton AC, Armstrong GG, Chipley PL. Pressurevolume curves of the arterial and venous systems in live dogs. Am J Physiol 1956; 184:253-8.
Sagawa K, Lie RK, Schaefer J. Translation of Otto Frank’s paper “Die Grundform des Arteriellen Pulses” Zeitschrift fur Biologie 37: 483-526 (1899). J Mol Cell Cardiol 1990; 22:253-4.
Westerhof N, Lankhaar JW, Westerhof BE. The arterial Windkessel. Med Biol Eng Comput 2009; 47:131-41.
Starling EH. The Linacre lecture on the law of the heart. In: Starling on the Heart: Facsimile Reprints, Including the Linacre Lecture on the Law of the Hear. London: Dowson of Pall Mall, 1965.
Greene DG, Carlisle R, Grant C, Bunnell IL. Estimation of left ventricular volume by one-plane cineangiography. Circulation 1967; 35:61-9.
Bunnell IL, Grant C, Greene DG. Left ventricular function derived from the pressure-volume diagram. Am J Med 1965; 39:881-94.
Suga H. Time course of left ventricular pressure-volume relationship under various end diastolic volume. Jpn Heart J 1969; 10:509-15.
Suga H. Time course of pressure-volume relationship of the cardiac ventricle in anesthetized dogs. Nihon Seirigaku Zasshi 1969; 31:328-9.
Suga H. Time course of left ventricular pressure-volume relationship under various extents of aortic occlusion. Jpn Heart J 1970; 11:373-8.
Sagawa K. The ventricular pressure-volume diagram revisited. Circ Res 1978; 43:677-87.
Suga H, Sagawa K. Mathematical interrelationship between instantaneous ventricular pressure-volume ratio and myocardial force-velocity relation. Ann Biomed Eng 1972; 1:160-81.
Suga H, Sagawa K, Shoukas AA. Load independence of the instantaneous pressure-volume ratio of the canine left ventricle and effects of epinephrine and heart rate on the ratio. Circ Res 1973; 32:314-22.
Sagawa K, Suga H, Shoukas AA, Bakalar KM. Endsystolic pressure-volume ratio: a new index of ventricular contractility. Am J Cardiol 1977; 40:748-53.
Suga H, Sagawa K. Instantaneous pressure-volume relationships and their ratio in the excised, supported canine left ventricle. Circ Res 1974; 35:117-26.
Sagawa K. Editorial: The end-systolic pressure-volume relation of the ventricle: definition, modifications and clinical use. Circulation 1981; 63:1223-7.
Sunagawa K, Maughan WL, Sagawa K. Effect of regional ischemia on the left ventricular end-systolic pressure-volume relationship of isolated canine hearts. Circ Res 1983; 52:170-8.
Mehmel HC, Schwarz F, Ruffmann K, Manthey J, von OK, Kubler W. End-systolic pressure-volume and endsystolic stress-volume relationships in patients with aortic stenosis and with normal valvular function. Basic Res Cardiol 1983; 78:338-50.
Maughan WL, Sunagawa K, Burkhoff D, Graves WL, Hunter WC, Sagawa K. Effect of heart rate on the canine end-systolic pressure-volume relationship. Circulation 1985; 72:654-9.
Sunagawa K, Maughan WL, Friesinger G, Guzman P, Chang MS, Sagawa K. Effects of coronary arterial pressure on left ventricular end-systolic pressure-volume relation of isolated canine heart. Circ Res 1982; 50:727-34.
Burkhoff D, de Tombe PP, Hunter WC. Impact of ejection on the magnitude and time course of ventricular pressure generating capacity. Am J Physiol 1993; 265 (HCP 34):H899-H909.
Burkhoff D, Oikawa RY, Sagawa K. Influence of pacing site on canine left ventricular contraction. Am J Physiol 1986; 251 (HCP 20):H428-H435.
Maughan WL, Sunagawa K, Burkhoff D, Sagawa K. Effect of arterial impedance changes on the end-systolic pressure-volume relation. Circ Res 1984; 54:595-602.
Burkhoff D, Sugiura S, Yue DT, Sagawa K. Contractilitydependent curvilinearity of end-systolic pressure- volume relations. Am J Physiol 1987; 252 (HCP 21):H1218-H1227.
Burkhoff D, Yue DT, Hunter WC, Sagawa K. End-Systolic Pressure-Volume Relationships (ESPVR) of Extrasystoles and Postextrasystoles From Iosolated Perfused Canine Ventricles (abstr). Circulation 1983;68 (IV):IV-1482.
Burkhoff D, Mirsky I, Suga H. Assessment of systolic and diastolic ventricular properties via pressure-volume analysis: a guide for clinical, translational, and basic researchers. Am J Physiol Heart Circ Physiol 2005; 289:H501-H512.
Senzaki H, Chen CH, Kass DA. Single-beat estimation of end-systolic pressure-volume relation in humans. A new method with the potential for noninvasive application. Circulation 1996; 94:2497-506.
Klotz S, Hay I, Dickstein ML, et al. Single-beat estimation of end-diastolic pressure-volume relationship: a novel method with potential for noninvasive application. Am J Physiol Heart Circ Physiol 2006; 291:H403-H412.
Westerhof N, Elzinga G, Sipkema P. An artificial arterial system for pumping hearts. J Applied Physiol 1971; 31:776-81.
Hay I, Rich J, Ferber P, Burkhoff D, Maurer MS. Role of impaired myocardial relaxation in the production of elevated left ventricular filling pressure. Am J Physiol Heart Circ Physiol 2005; 288:H1203-H1208.
Kaye D, Shah SJ, Borlaug BA, et al. Effects of an interatrial shunt on rest and exercise hemodynamics: results of a computer simulation in heart failure. J Card Fail 2014; 20:212-21.
Burkhoff D. HARVI: Cardiovascular Physiology & Hemodynamics. Part I. Basic Physiological Concepts (Version 2.0.0) [Mobile application software]. Retreived from https:// itunes.apple.com/gb/app/harvi/id568196279?mt=8. Last viewed on 11-20-2014.
Burkhoff D. HARVI: Cardiovascular Physiology & Hemodynamics. Part II. Advanced Physiological Concepts (Version 2.0.0) [Mobile application software]. Retreived from https://itunes.apple.com/gb/app/harvi/id568196279?mt=8. Last viewed on 11-20-2014.
Lumens, J., Delhaas, T., Reesink, K. D., Arts, T., and Dassen, W. Circadapt. Available Online at http://www.circadapt.org. Last viewed on 11-20-2014
Maughan WL, Shoukas AA, Sagawa K, Weisfeldt ML. Instantaneous pressure-volume relationship of the canine right ventricle. Circ Res 1979; 44:309-15.
Dickstein ML, Yano OJ, Burkhoff D, Spotnitz HM. Right Ventricular Contractile State Assessed With the Conductance Catheter and Sonomicrometry in the Pig (abstr). Anes Analg 1994;78:S79.
Alexander JJr, Sunagawa K, Chang N, Sagawa K. Instantaneous pressure-volume reltation of the ejecting canine left atrium. Circ Res 1987; 61:209-19.
Weimar T, Watanabe Y, Kazui T, et al. Differential impact of short periods of rapid atrial pacing on left and right atrial mechanical function. Am J Physiol Heart Circ Physiol 2012; 302:H2583-H2591.
Dickstein ML, Burkhoff D. A theoretical analysis of the effect of pulmonary vasodilation on pulmonary venous pressure: implications for inhaled nitric oxide therapy. J Heart Lung Transplant 1996; 15:715-21.
Hay I, Rich J, Ferber P, Burkhoff D, Maurer MS. The Role of Impaired Myocardial Relaxation in the Production of Elevated Left Ventricular Filling Pressure. Am J Physiol Heart Circ Physiol 2004.
Burkhoff D, Naidu SS. The science behind percutaneous hemodynamic support: a review and comparison of support strategies. Catheter Cardiovasc Interv 2012; 80:816-29.
Bartoli CR, Giridharan GA, Litwak KN, et al. Hemodynamic responses to continuous versus pulsatile mechanical unloading of the failing left ventricle. ASAIO J 2010; 56:410-6.
Kono S, Nishimura K, Nishina T, et al. Autosynchronized systolic unloading during left ventricular assist with a centrifugal pump. J Thorac Cardiovasc Surg 2003; 125:353-60.
Rupprecht L, Florchinger B, Schopka S, et al. Cardiac decompression on extracorporeal life support: a review and discussion of the literature. ASAIO J 2013; 59:547-53.
Combes A, Leprince P, Luyt CE, et al. Outcomes and longterm quality-of-life of patients supported by extracorporeal membrane oxygenation for refractory cardiogenic shock. Crit Care Med 2008; 36:1404-11.
Morley D, Litwak K, Ferber P, et al. Hemodynamic effects of partial ventricular support in chronic heart failure: Results of simulation validated with in vivo data. J Thorac Cardiovasc Surg 2007; 133:21-8.
Meyns B, Klotz S, Simon A, et al. Proof of concept: hemodynamic response to long-term partial ventricular support with the synergy pocket micro-pump. J Am Coll Cardiol 2009; 54:79-86.
Ewert P, Berger F, Nagdyman N, et al. Masked left ventricular restriction in elderly patients with atrial septal defects: a contraindication for closure? Catheter Cardiovasc Interv 2001; 52:177-80.
Sondergaard L, Reddy V, Kaye D, et al. Transcatheter treatment of heart failure with preserved or mildly reduced ejection fraction using a novel interatrial implant to lower left atrial pressure. Eur J Heart Fail 2014; 16:796-801.
Punnoose L, Burkhoff D, Rich S, Horn EM. Right ventricular assist device in end-stage pulmonary arterial hypertension: insights from a computational model of the cardiovascular system. Prog Cardiovasc Dis 2012; 55:234-43.
Dickstein ML, Spotnitz HM, Rose EA, Burkhoff D. Heart reduction surgery: an analysis of the impact on cardiac function. J Thorac Cardiovasc Surg 1997; 113:1032-40.
Artrip JH, Oz M, Burkhoff D. Left ventricular volume reduction surgery for heart failure: a physiologic perspective. J Thoracic Cardiovasc Surgery 2001; 122:775-82.
Gorcsan J, Feldman AM, Kormos RL, Mandarino WA, Demetris AJ, Batista RJV. Heterogeneous immediate effectsof partial left ventriculectomy on cardiac performance. Circulation 1998; 97:839-42.
Schreuder JJ, Steendijk P, van der Veen FH, et al. Acute and short-term effects of partial left ventriculectomy in dilated cardiomyopathy: assessment by pressure-volume loops. J Am Coll Cardiol 2000; 36:2104-14.
Starling RC, McCarthy PM, Buda T, et al. Results of partial left ventriculectomy for dilated cardiomyopathy: hemodynamic, clinical and echocardiographic observations. J Am Coll Cardiol 2000; 36:2098-103.
Franco-Cereceda A, McCarthy PM, Blackstone EH, et al. Partial left ventriculectomy for dilated cardiomyopathy: is this an alternative to transplantation? J Thorac Cardiovasc Surg 2001; 121:879-93.
Schreuder JJ, Castiglioni A, Maisano F, et al. Acute decrease of left ventricular mechanical dyssynchrony and improvement of contractile state and energy efficiency after left ventricular restoration. J Thorac Cardiovasc Surg 2005; 129:138-45.
Jones RH, Velazquez EJ, Michler RE, et al. Coronary Bypass Surgery with or without Surgical Ventricular Reconstruction. N Engl J Med 2009.
Ten Brinke EA, Klautz RJ, Tulner SA, et al. Long-term effects of surgical ventricular restoration with additional restrictive mitral annuloplasty and/or coronary artery bypass grafting on left ventricular function: six-month follow-up by pressure-volume loops. J Thorac Cardiovasc Surg 2010; 140:1338-44.
Burkhoff D, Wechsler AS. Surgical ventricular remodeling: a balancing act on systolic and diastolic properties. J Thorac Cardiovasc Surg 2006; 132:459-63.
Suga H. Ventricular Energetics. Phys Rev 1990; 70:247-77.
Fallick C, Sobotka PA, Dunlap ME. Sympathetically mediated changes in capacitance: redistribution of the venous reservoir as a cause of decompensation. Circ Heart Fail 2011; 4:669-75.
Dunlap ME, Sobotka PA. Fluid re-distribution rather than accumulation causes most cases of decompensated heart failure. J Am Coll Cardiol 2013; 62:165-6.
Semigran MJ, Cockrill BA, Kacmarek R, et al. Hemodynamic effects of inhaled nitric oxide in heart failure. J Am Coll Cardiol 1994; 24:982-8.
Kieler-Jensen N, Ricksten SE, Stenqvist O, et al. Inhaled nitric oxide in the evaluation of heart transplant candidates with elevated pulmonary vascular resistance. J Heart Lung Transplant 1994; 13:366-75.
Bocchi EA, Bacal F, Auler Junior JO, Carmone MJ, Bellotti G, Pileggi F. Inhaled nitric oxide leading to pulmonary edema in stable severe heart failure. Am J Cardiol 1994; 74:70-2.
Schreuder JJ, Maisano F, Donelli A, et al. Beat-to-beat effects of intraaortic balloon pump timing on left ventricular performance in patients with low ejection fraction. Ann Thorac Surg 2005; 79:872-80.
Jones RH, Velazquez EJ, Michler RE, et al. Coronary bypass surgery with or without surgical ventricular reconstruction. N Engl J Med 2009; 360:1705-17.
Burkhoff D, Maurer MS, Joseph SM, v. Left Atrial Decompression Pump for Severe Heart Failure with Preserved Ejection Fraction: Theoretical and Clinical Considerations. JCHF . 2014.
Sunagawa K, Maughan WL, Burkhoff D, Sagawa K. Left ventricular interaction with arterial load studied in isolated canine ventricle. Am J Physiol 1983;245 (HCP 14):H773-H780
Burkhoff D, Mirsky I, Suga H. Assessment of systolic and diastolic ventricular properties via pressure-volume analysis: a guide for clinical, translational, and basic researchers. Am J Physiol Heart Circ Physiol 2005;289:H501-H512
Baan J, van der Velde ET, de Bruin HG, Smeenk GJ, Koops J, van Dijk AD, Temmerman D, Senden J, Buis B. Continuous measurement of left ventricular volume in animals and humans by conductance catheter. Circulation 1984;70:812-823
Kass DA, Yamazaki T, Burkhoff D, et al. Determination of left ventricular end-systolic pressure-volume relationships by the conductance catheter. Circulation 1986;73:586-95
Alexander JJr, Sunagawa K, Chang N, Sagawa K. Instantaneous pressure-volume reltation of the ejecting canine left atrium. Circ Res 1987;61:209-219
Chapman CB, Ewer SM, Kelly AF, Jacobson KM, Leal MA, Rahko PS. Classification of left ventricular diastolic function using American Society of Echocardiography Guidelines: agreement among echocardiographers. Echocardiography 2013;30:1022-1031
Maughan WL, Sunagawa K, Sagawa K. Ventricular systolic interdependence: volume elastance model in isolated canine hearts. Am J Physiol (Heart Circ Physiol ) 1987;253:H1381-H1390
Bers DM. Cardiac excitation-contraction coupling. Nature 2002;415:198-205
Suga H. Ventricular Energetics. Phys Rev 1990;70:247-277
Weiss RG, Maslov M. Normal myocardial metabolism: fueling cardiac contraction. Adv Stud Med 2004;4:S457-S463
Duncker DJ, Bache RJ. Regulation of coronary blood flow during exercise. Physiol Rev 2008;88:1009-1086
Sunagawa K, Maughan WL, Friesinger G, Guzman P, Chang MS, Sagawa K. Effects of coronary arterial pressure on left ventricular end-systolic pressure-volume relation of isolated canine heart. Circ Res 1982;50:727-734
Floras JS. Sympathetic nervous system activation in human heart failure: Clinical implications of an updated model. J Am Coll Cardiol 2009;54:375-385
Burkhoff D, Tyberg JV. Why does pulmonary venous pressure rise following the onset of left ventricular dysfunction: a theoretical analysis. Am J Physiol 1993;265 (HCP 34):H1819-H1828
Santamore WP, Burkhoff D. Hemodynamic consequences of ventricular interaction as assessed by model analysis. Am J Physiol 1991;260 (HCP 29):H146-H157
Dickstein ML, Burkhoff D. A theoretical analysis of the effect of pulmonary vasodilation on pulmonary venous pressure: implications for inhaled nitric oxide therapy. J Heart Lung Transplant 1996;15:715-721
Kelsey R, Botello M, Millard B, Zimmerman J. An online heart simulator for augmenting first-year medical and dental education. Proc AMIA Symp 2002370-374
Hay I, Rich J, Ferber P, Burkhoff D, Maurer MS.Role of impaired myocardial relaxation in the production of elevated left ventricular filling pressure. Am J Physiol Heart Circ Physiol 2005;288:H1203-H1208
Morley D, Litwak K, Ferber P, Spence P, Dowling R, Meyns B, Griffith B, Burkhoff D. Hemodynamic effects of partial ventricular support in chronic heart failure: Results of simulation validated with in vivo data. J Thorac Cardiovasc Surg 2007;133:21-28
Punnoose L, Burkhoff D, Rich S, Horn EM. Right ventricular assist device in end-stage pulmonary arterial hypertension: insights from a computational model of the cardiovascular system. Prog Cardiovasc Dis 2012;55:234-243
Kaye D, Shah SJ, Borlaug BA, Gustafsson F, Komtebedde J, Kubo S, Magnin C, Maurer MS, Feldman T, Burkhoff D. Effects of an interatrial shunt on rest and exercise hemodynamics: results of a computer simulation in heart failure. J Card Fail 2014;20:212-221
We are greatly appreciative of the editing efforts of the following faculty:

Harvi-Student Translation Editors
Chinese: Yuhui Sun, MD, Kunlun He, MD, and Hanyu Zhang, MD
German: Karl-Philip Rommel, MD y Philipp Lurz, MD
Portuguese: Livia Melro, MD e Vinícius Zofoli, MD
Spanish: Valentina Pinzón, MD, Juan David Porras, MD y Leonardo Salazar, MD
Arabic: Anas Zaqut, Mahmoud Odeh, Hadeel Al Kayed, Anas Zayad, Dr. Ahmad Turk and Dr. Kais Balbissi
French: Alexandre Mignon, MD
Ukrainian: Sviatoslav Kalashnikov, MD

Harvi-ECMO/MCS Translation Editors
Chinese: Yuhui Sun, MD, Kunlun He, MD, and Hanyu Zhang, MD
German: Karl-Philip Rommel, MD and Philipp Lurz, MD
Korean: Kim Ji-eon, MD, Lee Seung-hyung, MD, Jeong Jae-seung, MD
Japanese: Shingo Ichiba, MD PhD, Yu Hohri, MD, PhD, and Yuji Kaku, MD
Portuguese: Livia Melro, MD and Vinícius Zofoli, MD
Spanish: Leonardo Salazar, MD
Italian: Valentina Stella, MD and Matteo DiNardo, MD
Arabic: Lubna Alnatour, Rahmeh Al-Asmar, Mohammad Al-Rababa’h, Osama Maher Mustafa, Nour Hazem Mash’al, Siya J. Suwan

Harvi-ECMO/MCS Translation Editors
Chinese: AI Translation
French: AI Translation
German: AI Translation
Italian: AI Translation
Japanese: AI Translation
Korean: AI Translation
Portuguese: AI Translation
Spanish: AI Translation
These versions have been edited. If you find errors in these versions, please take a screen shot showing the error and send to: info@HarviDynamics.com
We are in the process of completing editing of
Harvi-Student: Italian, Korean, Japanese.
Harvi-ECMO/MCS: French
The machine translations have been posted and will be replaced by the edited versions as soon as they are compete.